Client background and why HIFU
The client came to the Toowong clinic having noticed, over two years, a softening along her jawline and through her mid-face. She had ruled out filler (she did not want added volume, she wanted support for her skin’s own structure) and arrived already familiar with energy-based treatments, asking specifically about HIFU. At consultation, her Registered Nurse assessed skin quality and degree of laxity and placed her within the mild-to-moderate range that responds to Ultraformer 3. Significant jowl descent sits outside the device’s scope and is appropriately referred for a specialist opinion.
Ultraformer 3 delivers focused acoustic energy to a precise focal point beneath the skin, creating controlled thermal zones that the body treats as micro-injuries. It is designed to stimulate collagen remodelling at SMAS depth and supports skin firmness over the following 3–6 months. It helps improve the appearance of mid-face laxity in cases like hers and is not a substitute for surgical intervention where indicated. The clinician was explicit at consultation: HIFU produces no visible change on the day of treatment. Any improvement develops gradually as new collagen forms.
Protocol
Topical anaesthetic was applied 30–45 minutes before the session. A combination of cartridge depths was used (deeper along the jawline and lower face to reach the SMAS, shallower through the mid-face for dermal layers) across mid-face, jawline and submental area. Total treatment time was approximately 60 minutes, with brief pauses for comfort over bony regions.
Phase-by-phase journey
At Week 0, the treated zones showed mild, diffuse pinkness that resolved within hours; no lifted appearance was visible, and clinician notes recorded “no significant visible lift at Week 0, expected.” Through Weeks 1–2 the client experienced transient tenderness along the jawline without bruising, returned to work the same day and reinforced daily SPF 50+ for the Brisbane climate. By Weeks 4–6, early textural firmness was noted on palpation at clinical review, though no photographic change was yet documented. At Week 12 the primary review frame showed a softening of the jawline shadow and a small but visible improvement in the mid-face contour compared with baseline. At Week 24 (within the expected 3–6 month window for collagen remodelling to settle), the review frame showed continued support for skin firmness along the jawline and submental area. Individual results vary.
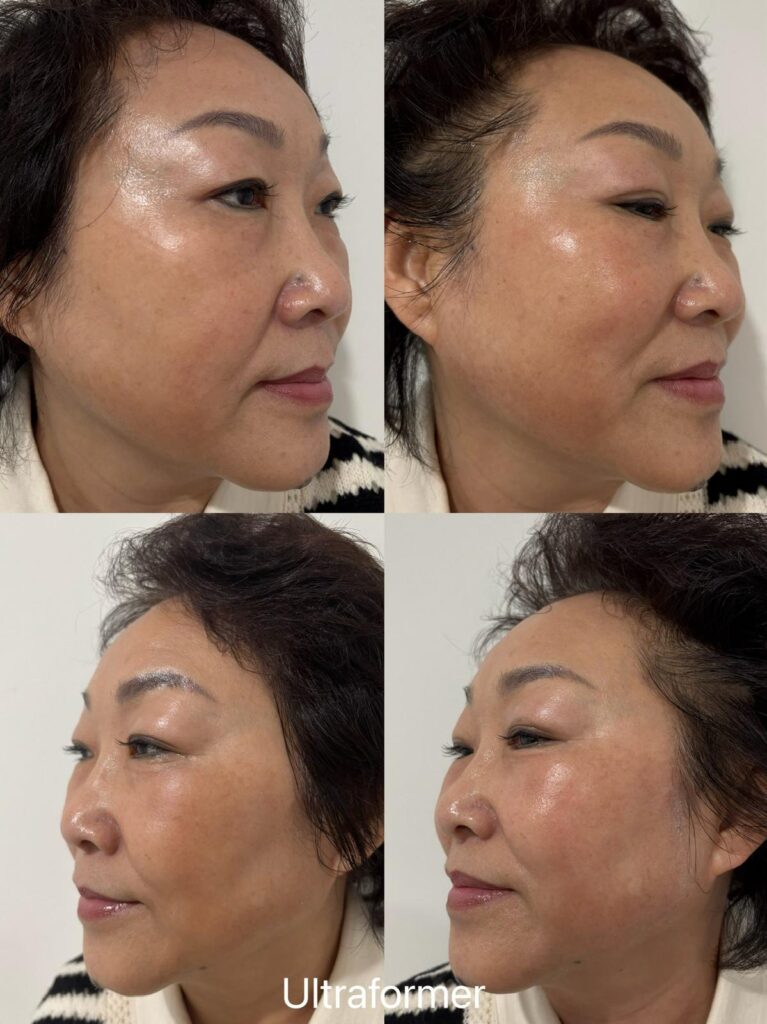
Maintenance
HIFU results are typically maintained for 12–18 months after a single session, with variability by baseline, age and lifestyle. This client is booked for a 12-month review to assess whether a maintenance session is indicated. Day-to-day, she was counselled on daily broad-spectrum SPF 50+, a well-tolerated topical routine, and adequate sleep and hydration: the conditions under which new collagen is best supported.
What this means for others
This describes one client in the mild-to-moderate laxity range with otherwise good baseline skin quality; it does not predict any other client’s response. Anyone considering HIFU should plan for a 3–6 month review window, accept that the sensation during treatment is real, and understand that it is not a substitute for surgical intervention where indicated. Suitability is determined at clinical consultation at Flourish Skin and Laser Clinic, Toowong.
Individual results vary.
Important information about this case study
Individual results vary. The images on this page show one client’s response to a specific treatment plan delivered at Flourish Skin and Laser Clinic. Your skin, concern, and response to treatment will be different.
Treatment performed: Ultraformer 3 HIFU, a non-invasive focused-ultrasound treatment designed to stimulate collagen remodelling at SMAS depth, supporting skin firmness over the following 3–6 months.
Clinician: Registered Nurse, Flourish Skin and Laser Clinic (Toowong).
Common side effects disclosed at consultation include: transient redness, mild swelling, tenderness in treated areas for a few days to 1–2 weeks, tingling or short-lived numbness, occasional small welts along treatment lines, and rarely bruising, temporary nerve irritation, or changes to fat distribution in the treated area. A full risk profile is reviewed with you in person before any treatment begins.
Suitability for Ultraformer 3 HIFU is determined during a clinical consultation. This case study is not a recommendation, diagnosis, or clinical guarantee. It is a factual record of one client’s treatment course, published with their written consent. HIFU is not a substitute for surgical intervention where indicated.
Device / product regulatory status: Ultraformer 3 is manufactured by Classys, Inc. (Korea), supplied in Australia by Classys Australia Pty Ltd, and included on the Australian Register of Therapeutic Goods as Focused Ultrasound Therapeutic Equipment (ARTG 410730). [CLINIC TO CONFIRM: any model-specific ARTG ID distinct from 410730.]
Photo conditions: Both photographs on this page were taken under matched lighting, distance, angle, and device. No filters, beauty smoothing, or retouching have been applied. Original files retained on record.
Consent and withdrawal: This case study is published with the client’s specific written consent for use on the Flourish Skin and Laser Clinic website only. The client may request removal at any time by contacting enquiry@fskinandlaser.com.
Book a consultation at Toowong to discuss whether Ultraformer 3 HIFU is appropriate for your skin.